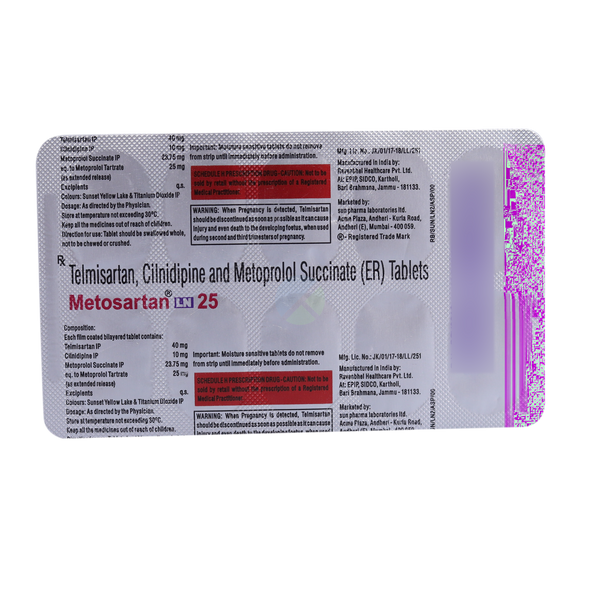
Description
Metosartan LN 25 Tablet ER is a prescription medicine used to control high blood pressure (hypertension). It combines three active ingredients:
-
Telmisartan (40mg): An angiotensin receptor blocker (ARB) that relaxes blood vessels to improve blood flow.
-
Metoprolol Succinate (23.75mg): A beta-blocker that lowers heart rate and reduces cardiac workload.
-
Cilnidipine (10mg): A calcium channel blocker (CCB) that dilates blood vessels and provides smooth blood circulation.
Together, this combination helps in effectively lowering blood pressure, reducing strain on the heart, and preventing complications like stroke, heart attack, and kidney damage.
Uses:
-
Treatment of high blood pressure
-
Prevention of heart attack and stroke
-
Protection of kidney and heart health in hypertensive patients
How to Use:
-
Take the tablet as advised by your doctor, once daily.
-
Swallow whole with a glass of water; do not crush or chew.
-
Can be taken with or without food, but at the same time every day.
Dosage:
-
Dose is determined by the doctor based on medical condition and response to therapy.
-
Do not miss doses or stop suddenly without medical advice.
Precautions:
-
Inform your doctor if you have heart, kidney, or liver disease.
-
Avoid alcohol as it may enhance dizziness or low blood pressure.
-
Not recommended in pregnancy and breastfeeding without doctor’s advice.
-
Regular blood pressure and heart rate monitoring is recommended.
Possible Side Effects:
-
Dizziness, headache, or fatigue
-
Slow heartbeat (bradycardia)
-
Low blood pressure
-
Swelling of ankles or feet (edema)
-
Flushing or warmth
-
Stomach upset or nausea







Leave a Reply