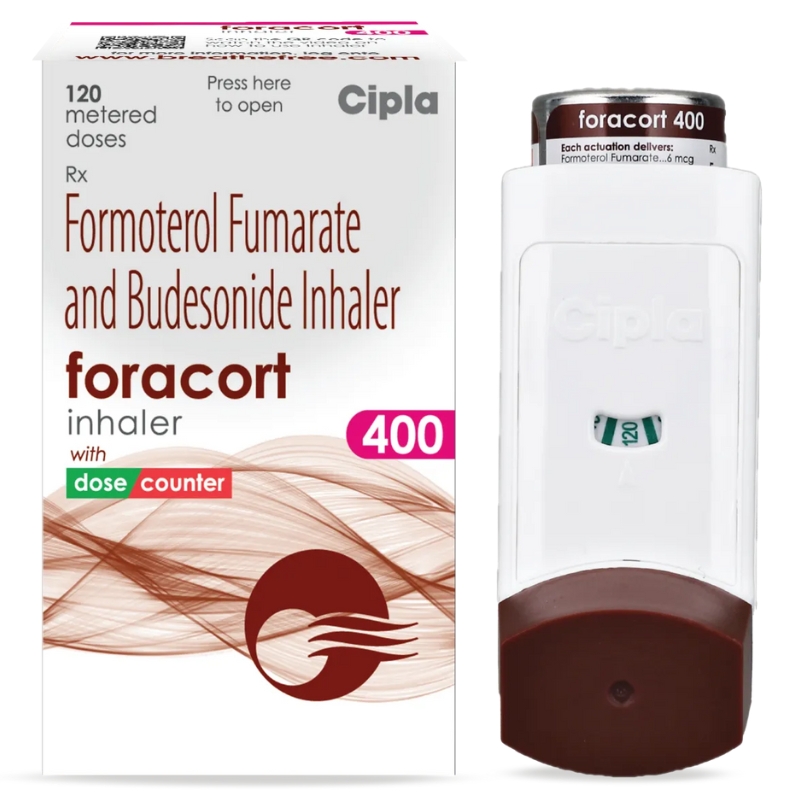
Description
FORAcort Inhaler 400 is a combination inhaler used for the long-term management of asthma and chronic obstructive pulmonary disease (COPD). It contains Formoterol, a bronchodilator that relaxes airway muscles for easier breathing, and Budesonide, a corticosteroid that reduces inflammation and swelling in the lungs. This dual-action formula helps prevent wheezing, coughing, and shortness of breath, improving overall lung function and quality of life.
Uses:
-
Maintenance treatment of asthma
-
Control and prevention of COPD symptoms
-
Reduces airway inflammation and improves breathing efficiency
How to Use:
Shake the inhaler well before use. Breathe out fully, place the mouthpiece in your mouth, and press the inhaler while inhaling deeply and slowly. Hold your breath for about 10 seconds before exhaling. Rinse your mouth thoroughly with water after each use to prevent fungal infections such as oral thrush.
Dosage:
The usual recommended dose is 1–2 puffs twice daily or as directed by your physician. Do not exceed the prescribed dose or stop using suddenly, as it may worsen your breathing condition.
Precautions:
Use with caution if you have heart disease, high blood pressure, thyroid disorders, diabetes, liver problems, or tuberculosis. Not for relief of acute asthma attacks — use a rescue inhaler in emergencies. Consult your doctor before use if pregnant or breastfeeding.
Side Effects:
Common side effects include throat irritation, hoarseness, cough, nausea, tremors, headache, and oral fungal infection. To reduce side effects, rinse your mouth after each use. Seek immediate medical help if you experience chest pain, irregular heartbeat, or worsening breathing difficulty.






Leave a Reply